High-tech manufacturing requires rigorous precision cleaning processes because contamination control determines the life or death of today’s highly sophisticated devices. Products used in the semiconductor industry, for example, require precision cleaning to meet stringent performance criteria.
There can be numerous steps involved to achieve the quality standards and guaranteeing cleanliness is essential. The same is true for printed circuit boards (PCBs), which are now smaller and more densely populated. PCBs must leave the production line spotlessly clean. Poor cleaning degrades inspection, fault detection, product performance and longevity. Therefore, high-tech device manufacturers should consider cleaning a mission-critical process. Without rigorous cleaning processes in place, the outcome for high-tech devices can be detrimental.
Selecting an effective cleaning chemical is critical to the life of PCBs to prevent age-related failures such as shorting caused by dendritic growth or poor conformal coating adhesion.
With so many options on the market, finding a solution to clean highly complex devices can be bewildering. Fortunately, there is a formula to help in predicting the effectiveness of a cleaning fluid: the wetting index. It provides an indication of how well the cleaning fluid will be able to flow under and around tightly packed components to lift and remove debris away from the surface.
Better wetting equals better cleaning
‘Wetting’ refers to a liquid’s ability to remain in contact with a surface. This is important with high-tech manufacturing because if a cleaning fluid cannot penetrate between closely spaced parts to remove contamination, then malfunction is likely. Therefore, better wetting will result in better cleaning.
Particulate and insoluble debris can be lifted and removed by high-density fluids; however, the fluid needs to also have appropriate viscosity and surface tension to flow in and out of those tight spaces. Fluids with low wetting indexes (high surface tension and viscosity) will not be able to flow into constricted spaces to remove trapped contamination.
The most important score is the surface tension. Fluids with low surface tension will clean better than those with higher surface tension
The wetting index combines these three different, but relevant, chemical characteristics (density, surface tension and viscosity) into a single score. The total helps to predict the quality of the fluid as a cleaning agent.
The most important score is the surface tension. Fluids with low surface tension will clean better than those with higher surface tension. Water is a prime example. Water can be a good cleaner for many types of contamination, but it has a high surface tension and a higher viscosity than many cleaning chemistries.
These properties make it more difficult to flow in and out of the tight spaces found in microelectronics. It can become a problem when water gets into a tight space but then is unable to find a way back out. The result is that all the contamination that the water just moved is now trapped in a space that cannot be easily accessed. Fluids with lower surface tensions and viscosities are more able to move in and out of these constricted spaces and prevent contamination from getting trapped.
Avoid the pitfalls of cleaning
Viscosity is an important calculation within the wetting index. It is a chemical characteristic that describes a fluid’s ability to flow quickly, smoothly and without outside effort. It is defined as a liquid’s resistance to gradual deformation. This corresponds to the informal concept of “thickness”. It is an indirect measure of the molecular attraction within a fluid for itself; highly viscous fluids are strongly attracted to their own molecules and resist separation.
The viscosity of water, or other inefficient cleaners like Isopropyl Alcohol (IPA), works against good cleaning. Higher viscosities mean the fluid moves sluggishly and oppose deformation resisting wetting the surface of all the parts to be cleaned. It will not move into tight spaces easily, and if water gets into small crevices, it may not come out, resulting in your cleaning efforts being ineffective.
Modern cleaning chemistries are heavier; typically 20%-30% heavier than water, and 50%+ heavier than alcohol. This means it can get under particulate and easily lift it off the surface. Remember the lower the viscosity score, the better the cleaning result.
How does aqueous cleaning score?
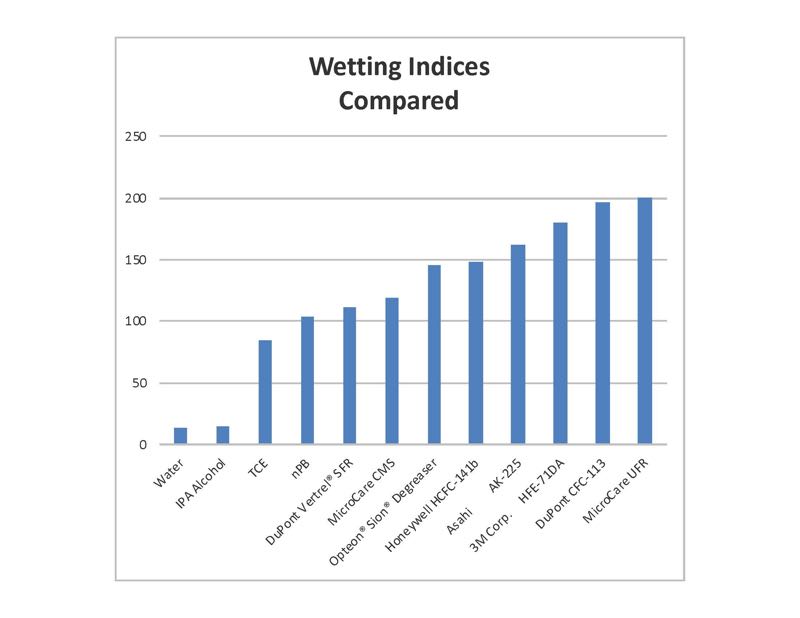
As demonstrated above, the cleaning performance of water is often not adequate for today’s modern high-tech devices. The surface tension of water is the highest of any cleaning agent available, this results in a low wetting index; a score of only 14.
When compared to highly-advanced non-flammable cleaning fluids, which on average score above 100, it is clear to see why they are winning in the fight against contamination as PCBs get smaller and more complex.
The very poor wetting index of water is a function of its inherent molecular structure, which cannot be changed
Water-based cleaning was once the favoured process, but perceptions are changing as manufacturers migrate to specialised cleaning fluids.
The very poor wetting index of water is a function of its inherent molecular structure, which cannot be changed. This means modern, environmentally friendly fluids will out-perform water because they simply ‘wet’ better. This results in quicker, more effective and more flexible cleaning of small, delicate or intricate shapes.
While PCBs are complicated devices with complex shapes, remember that ‘wettability’ concerns should be considered even for relatively flat surfaces. Cleaning agents with enhanced wettability will be better able to penetrate layers of soil, dissolving residues and lifting particles from the surface of even flat substrates.
By combining density, viscosity and surface tension scores into a wetting index, it simplifies comparisons into a single number. In general, the higher the score, the better the cleaning.
The formula for working out a wetting index is:
(Density x 1000)
________________
(Surface Tension x Viscosity)
To improve the wetting index score of aqueous-based cleaners a surfactant can be added to lower the surface tension. But even with the addition of surfactants, the surface tension will still be twice that of any modern cleaning fluid.

Working out a wetting index score will give a more explicit indication of a cleaner’s ability to do its job well. By using a simple calculation, the correct solution will be selected to clean effectively.
There is no doubt in that cleaning correctly is key to contamination control in high-tech cleanroom environments, and the wetting index is an ally to that, as determining the score will help in assessing a fluid’s ability to penetrate tight spaces and leave them uncontaminated.
When scoring a cleaning solution also investigate its solubility, this is normally measured with pH for water-based cleaners and kauri-butanol (Kb) value for solvents. Cleaning solutions can be tailored to have a wide range of Kb values, which gives them a very versatile cleaning performance. Don’t forget to investigate other factors including drying speed, aroma, toxicity, flammability and other concerns to get an overall picture of a cleaner’s ability to dissolve or remove the target contamination.
There are many techniques to work out just how effective a cleaning fluid will be. The wetting index can be a significant parameter in determining the best cleaning fluid for an application and a good starting block.
Sorting through the wide variety of cleaning solutions can be a tiring task; the wetting index is just one more datum to help narrow down the options to identify a viable solution for any critical cleaning process.
Reference
Parts Cleaning: Does the Cleaning Agent Work? Barbara Kanegsberg & Ed Kanegsberg, BFK Solutions




