Biopharmaceutical manufacturers are expanding their global footprints and capacity to meet new drug and market demands. One way they are achieving this growth is through single-use systems (SUS).
Single-use production enables a more flexible, cost-effective approach to biopharma manufacturing and has been adopted rapidly into cGMP manufacturing processes such as upstream, downstream and fill-and-finish process steps.
However, with the rapid growth of single-use production, especially by drug manufacturers with multiple locations across the globe, there are risks associated with underestimating the regulatory compliance, manufacturing and quality requirements of these systems.
Using SUS also introduces new logistics challenges that, if not correctly understood and planned for, can leave biopharma manufacturers vulnerable to supply chain complexities.
Any disruption to the supply chain may result in devastating effects to biologic production and, ultimately, to patients using the drugs. To minimise these risks, biopharmaceutical manufacturers can benefit by adopting a global single-use supply chain strategy early in the drug development lifecycle. Such an approach can help prevent potential disruptions that could hamper product manufacturing and regulatory approvals.
Regulatory landscape
Drug manufacturers that rely on single-use equipment face an increasingly rigorous quality and regulatory process. They need to manage critical raw material suppliers, conduct site and customer audits and solve the challenges associated with engineering and manufacturing unique, custom-designed therapies.
Product sterility validation that follows a recognised reference standard, identified by regulations such as ANSI/AAMI/ISO 11137, should be investigated. This is commonly referred to as VDmax25
When reviewing a prospective supplier of SUS, there are two primary considerations for biopharma manufacturers to keep in mind to help mitigate their supply chain risks: the regulatory and quality compliance and the supply chain operational excellence of the supplier.
Several main criteria for regulatory and quality compliance are worth considering when choosing a supplier for single-use manufacturing products and technology.
First, the environment where the single-use equipment and materials are being manufactured should be checked to ensure proper controls are in place to monitor and track environmental conditions where the materials are produced. These procedures are commonly referred to as cleanroom management.
A typical cleanroom for SUS manufacturing will be specified as an ISO Class 7 (Class 10,000) cleanroom. The production facility should have documented evidence of annual cleanroom recertification and a validated process showing a rotational cleaning regimen, as well as air/surface viable and nonviable particulate testing being carried out on a scheduled basis.
Cleanrooms should also be monitored for real-time temperature, humidity and differential pressure with defined action and alert criteria.
Also, product sterility validation that follows a recognised reference standard, identified by regulations such as ANSI/AAMI/ISO 11137, should be investigated. This is commonly referred to as VDmax25.
VDmax25 assures that, if something is gamma irradiated to 25 kGy, it has a sterility assurance level of 10-6; a 1/1,000,000 chance that a living organism will be found after the gamma process is performed.
The validation, however, should not be a single-point one; there should be documented evidence that periodic revalidation is performed on a routine basis as defined by the standard and the single-use supplier.
A sterile barrier packaging validation is also used to establish shelf life for an irradiated product, which is different from sterility validation.
The single-use supplier’s ability to perform lot release testing on finished products, to verify that the product meets specific requirements after sterilisation, should be the third item reviewed.
Lot release testing is a relatively new requirement that has been driven by downstream applications after filtration. Most requests for this testing include USP <85> for bacterial endotoxins and USP <788> for particulate contamination.
Quality risk management
The fourth, and potentially most important item for reducing risk in the single-use supply chain, is to fully understand a supplier’s quality risk management (QRM) programme.
It is critically important that single-use suppliers adopt a systematic process for the assessment, control, communication and review of risks to the quality of manufactured products.
A key element of ISO 9001 and cGMP related to quality is risk-based decision-making, and a qualified single-use supplier will have developed quality metrics to control their manufacturing risks.
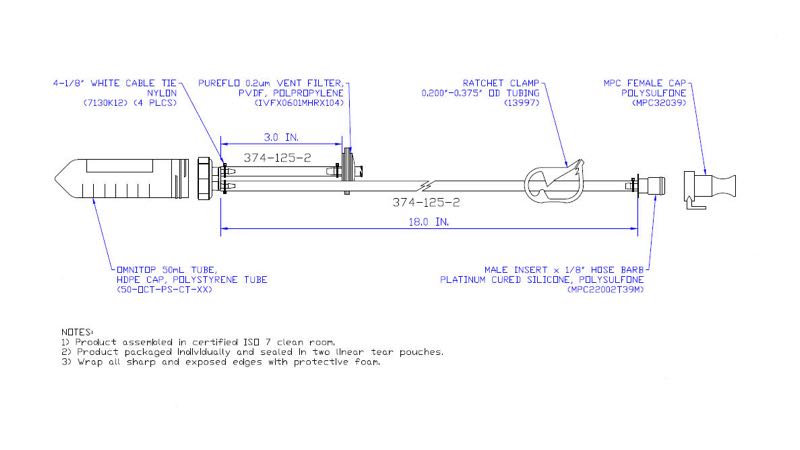
Quality metric and monitoring indicators may include on-time delivery, turnaround time for engineer’s drawings, quality documentation turnaround and tracking of manufacturing defects
Essential steps in risk management generally include risk assessment (identification, analysis and evaluation); risk control (mitigation, reduction and acceptance); and risk review.
It’s helpful to consider several key points within a supplier’s QRM, such as their development of a risk register: a senior level management evaluation tool used to identify and review where the greatest risks may occur. This document can change over time as risks are identified and mitigated.
Quality metric and monitoring, a second key element of a QRM, shows ongoing improvement and tracks important quality indicators.
Best practices should include developing a risk-based classification of raw material suppliers, performing routine audits of those high-risk suppliers, establishing quality agreements and performance reports
For a single-use supplier, these may include on-time delivery, turnaround time for engineer’s drawings, quality documentation turnaround and tracking of manufacturing defects.
Single-use suppliers should establish a quality goal and then track performance and perform trend analysis to respond to and improve their processes.
A third QRM function is supplier quality management. Conducting risk-based audits of raw material suppliers gives single-use products and systems providers the ability to understand the quality management capabilities of new companies that seek to provide raw materials.
With this insight and data, single-use suppliers can collaborate effectively with raw material providers to promptly address issues and implement corrective and preventative actions.
Best practices should include developing a risk-based classification of raw material suppliers, performing routine audits of those high-risk suppliers, establishing quality agreements and performance reports, and tracking this performance in some measurable, actionable process.
Finally, the fourth element of a proper QRM is seeing evidence of a business continuity plan.
Single-use suppliers should conduct a business impact analysis (BIA) to evaluate each potential scenario associated with business continuity and disaster recovery, so they are ready to respond if an event has the potential to interrupt operations.
Each scenario can be given a grade level related to both likelihood and impact: low, medium or high risk. Preventive measures and/or recovery objectives can then be identified for all scenarios with both a high impact and/or high occurrence rating. Scenarios with lower impact or likelihood potentials should also be evaluated on a case-by-case basis.
Supply chain OpEx
Managing risk and minimising the complexity of the global supply chain for single-use products and materials is supremely critical. Biopharma manufacturers should conduct an ongoing evaluation of a single-use supplier’s supply chain OpEx capabilities. Understanding and aligning these capabilities with the manufacturer’s own quality practices is important as biopharma manufacturers design their facilities entirely with SUS and expand globally.
Suppliers of single-use products and systems need to work collaboratively with their biopharmaceutical producer customers in planning, forecasting and replenishment to protect the biopharmaceutical manufacturer’s ability to effectively support sales and operational planning and keep orders delivered on time.
Supply chain operational excellence capabilities can be built around a four-step collaborative planning, forecasting and replenishment (CPFR) programme that includes:
- Understanding customer requirements such as important product/order attributes of dating, documentation, order frequency, lot control and storage-material handling requirements.
- Effectively transferring documented requirements to internal systems to “operationalise” things such as customer-care instructions, warehouse instructions, and setup of customer-specific inventory reserves.
- Regularly engaging in customer-planning meetings to obtain updated forecasts.
- Engaging with customers and suppliers to manage changes in key factors such as required components and lead times.
OpEx capabilities. Aligning these capabilities with the manufacturer’s own quality practices is important as biopharma manufacturers design their facilities entirely with single-use systems and expand globally
Supply chain assurance can also be achieved through a customer-centric approach with flexibility and solutions that decrease complexity around storage and delivery.
One way to achieve this is through sales and operations planning (S&OP), an integrated business management process that helps ensure the executive, or leadership team of an organisation, is continually focused and aligned across all functions.
The S&OP process includes an updated forecast that leads to a: sales plan; production plan; inventory plan; customer lead time (backlog) plan; new product development plan; strategic initiative plan; and resulting financial plan.
Plan frequency and planning horizon depend on the specifics of the industry. The short product lifecycles and high-demand volatility often associated with single-use products require a tighter S&OP than steadily consumed products. Done well, the S&OP process enables effective supply chain management.
Collaborative approach
One of the more challenging issues facing the single-use industry is faster lead times for finished goods. One way to mitigate the challenge of quicker lead times is to stock more finished goods, but this also has inherent risks related to proper warehousing and storage.Many biopharmaceutical manufacturers do not have the adequate warehousing infrastructure necessary to store large volumes of single-use products. They must seek alternate solutions for local storage and quick delivery.
Biopharmaceutical manufacturers with multiple operations around the world can help ensure their single-use production operations remain secure by working with single-use suppliers who combine ingenuity and product leadership with powerful channel and supply chain operations.
Ultimately, this should include superior single-use expertise with a collaborative approach in designing solutions; access to a broad product portfolio with components sourced from multiple suppliers; unique capabilities, such as expedited system design and delivery of drawings; and, ultimately, supply chain OpEx capabilities that are fully supported by regulatory and quality compliance.
N.B. This article is featured in the December 2018 issue of Cleanroom Technology. The digital edition is available online.




