The water system is one of the most basic utilities for every pharmaceutical production plant and its products—purified water (PW) and water for injection (WFI)—have an important part in almost every process. Hence, designing a reliable water production system of high-quality standard is critical.
Published last June, the new ISO 22519 “Purified water and water for injection pretreatment and production systems” has been written to improve reliability, standardise systems and maximise quality.
The standard is a comprehensive set of guidance rules with three main principles at its core:
- Hot water sanitisation
- Stainless steel construction
- Continuous bioburden reduction (CBR)
These principles acting together improve the microbiological levels in the water. Regular hot water sanitisations are a sure-fire way of controlling the incoming bacteria and stopping the development of biofilm. Stainless steel is a reliable material with little need of maintenance, with both piping and contractors regularly available all over the world. Continues bioburden reduction is achieved by selecting the appropriate process technologies, equipment and operational strategies, all commonly available on the global market.
While designing a new PW/WFI water system, or an upgrade, the above three principles must be implemented to meet the qualifying criteria of the ISO standard.
Hot water sanitised systems
All water system pretreatment has to keep the reverse osmosis (RO) membranes free of scale and needs to remove chlorine from the RO feed. In addition, pretreatment for PW and WFI systems also need to keep the whole system free of microbial growth and biofilm. The new ISO 22519 for water systems defines the three principles that are instrumental in achieving the microbial criteria and avoiding biofilm growth.
The standard states that “all parts of the PW/WFI pretreatment and production shall be hot water sanitised, from the feed water inlet valve to the PW/WFI fill valve”.
The hot water sanitisation principle is based on the Questions and Answers document released by the European Medicines Agency (EMA). The report describes the specific agents that can be used as part of a control strategy. In response to the question "What specific agents can be used as part of a control strategy?" it states: “Use of thermal sanitisation where possible should also be considered.” The temperature is set at 80ºC, above the recommended minimum of 75ºC, so as to make sure that even if the sanitisation is short, its effectiveness is not impaired.
According to the standard, chemical sanitisation such as chlorine addition is not enough for regular sanitisation, since it is not always effective against biofilm.
The following are key considerations for performing hot water sanitisation:
- The hot water cycle should be developed to include different equipment and piping
- The temperature and cycle time must be such the coldest point must be kept at minimum sanitisation temperature for over 30 min
- The sanitisation should be conducted at a minimum of 80°C at all points, during the sanitisation high-temperature hold
- All the components and instrumentation in the pre-treatment and production system should be compatible with hot water sanitisation
- The sanitisation parameters should be controlled and recorded
The standard also states that "all components in the PW/WFI pre-treatment and production system shall be manufactured from stainless steel (SS) 316/316L. All PW/WFI contact parts to be fabricated from SS 316L, including piping, tubing, tanks, pumps, heat exchanger, valves, instruments and other accessories”.
Construction material
The guidance insists on stainless steel construction, as it allows unlimited cycles of hot water sanitisation from the feed water inlet valve to the PW/WFI fill valve.

Stainless steel construction allows unlimited cycles of hot water sanitisation
Polymer-based heat resistant piping exists and is used in some parts of the world. This type of piping has not been included in the standard as both the material and the contractors are not always commonly available. As the ISO 22519 is a global standard, thoughts have been given to the fact that fabrication must also be globally available.
One of the main problems of installation of plastic-based piping is being able to achieve reliable welds with no beads. Short piping accessories with minimum dead leg are also available, but are expensive and not commonly in stock.
Continues bioburden reduction (CBR)
According to ISO 22519, the water quality must show an improvement in quality as the water advances through the system stages. The standard indicates specifically the following three parameters: microbial count, conductivity and total organic carbon (TOC).
The continuous bioburden control principle obligates to control bacterial growth from the pre-treatment stage. This is to prevent a high bacterial load on the reverse osmosis (RO) membrane that could lead to bacterial growth and eventually reach out of specification results.
As it appears in the standard (see table 1), the microbial total count at the RO feed should be <500 (CFU/ml); and after the RO should be <200 (CFU/ml).
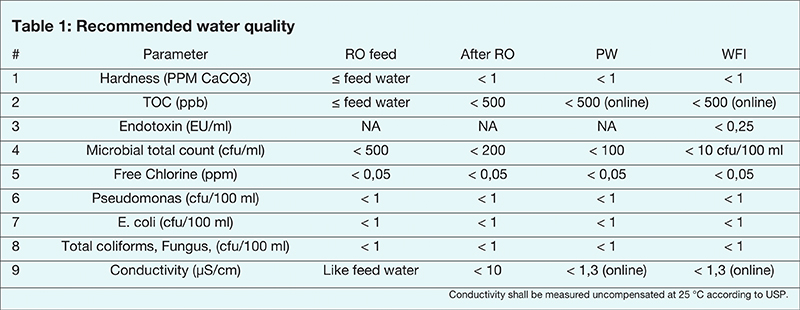
To comply with the continuous bioburden reduction principle, the following has been stipulated in the standard:
- The water production system should operate constantly 24/7, without stoppage of pump or flow, to retard microbial growth
- Hot water sanitisation should be performed from the raw water feed to the system and up to and including the PW/WFI feed to the tank or consumer
- Appropriate equipment that supports that CBR principle. Best to keep away from media-based softeners and high growth carbon filters
The new ISO 22519 “Purified water and water for injection pre-treatment and production systems” provides a benchmark that can be used by those that design, maintain, inspect or use PW/WFI water systems.
Anyone following the ISO 22519 principles will improve the reliability and will meet the microbial challenge better. The standard allows authorities and regulatory bodies to perform professional audits with a common checklist, and most importantly, it lets end-users to specify their water systems without being an expert, and to decide whether the water systems that were offered to them are safe, efficient and sustainable.
N.B. This article is featured in the August 2019 issue of Cleanroom Technology. The latest digital edition is available online.




