There are many factors that affect hydrogen peroxide (H2O2) biodecontamination efficacy. These factors include how the agent is generated and used; whether it is best as vapour, aerosol, wet, dry, semi-dry; concentration in the target space; and the prevailing process environmental conditions including temperature, relative humidity, and air movement. (Figure 1)
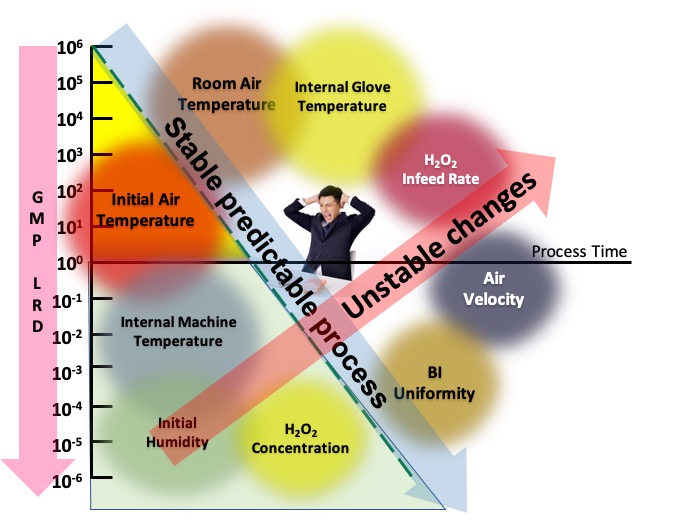
Impact of variables on H2O2 Bio-decontamination
These complex multi-factorial influences affect the relationship between deactivating bacterial spores and the contact between hydrogen peroxide on the target surface. Some of the challenges with traditional hydrogen peroxide biodecontamination were highlighted in April 2018 when Andrew Hopkins wrote on the UK MHRA blog a piece entitled “VHP (Vapour Hydrogen Peroxide) Fragility”.
This article and the all the ongoing debates initiated Airex’s search for, research and development of a much more robust biodecontamination process.This article outlines a new process, discovered, and developed by Airex that generates, injects, and disperses an exceptionally fine hydrogen peroxide aerosol into a closed environment under an ultrasonic reverberation energy field. This field causes the hydrogen peroxide aerosol to be distributed throughout the enclosure or chamber without the need for dispersion fans or other air movement. It helps to ensure formation of a uniform solution wetting layer on the target surface, leading to more reliable and faster biodecontamination. Experiments show that with this method, using relatively small amounts of solution, can achieve an almost uniform biodecontamination for different surface materials with short cycle times compared to the most found flash evaporation method (FE method). In this article, the performance will be described by comparing the new ultrasonic biodecontamination method (UD method) and the FE method and will consider the impact of temperature and relative humidity, the impact of air velocity over target surfaces, and the influence of surface materials.
During the investigation and development of this new technique, the use of traditional biological indicators (BI) was considered for the biodecontamination cycle development work but were rejected in favour of Protak enzymatic indicators (EI). These were preferred due to advantages of speed (minutes versus days); avoidance of so-called rogue BIs; and less indicator variability.
Some limitations of the FE method
The FE method is used in many applications worldwide and was used as a reference point or benchmark for developing and assessing the performance of the UD method. To establish this benchmark, the research team investigated the relationship between efficacy, temperature and relative humidity using log reduction (LRD) of EIs. These studies identified that the multivariate design space was extremely complex, principally due to the temperature difference between the vapour/air mixture and critical surfaces inside an isolator enclosure for example.
During an FE method cycle, the temperature inside the isolator chamber always rises gradually, and the temperature of internal surfaces are not always constant because of variations of specific heat capacity and heat transfer affected by boundary layer thickness and related air velocity. The effect of these temperature changes and differences cause the micro-condensation of hydrogen peroxide solution on the target surfaces to be variable and unstable.
Characteristics of the UD Method
The UD Method comprises an ultrasonic aerosol generator with an associated ultrasonic aerosol diffuser (Figure 2). The generator generates the primary aerosol, and the diffuser propels and helps diffuse the fine aerosol throughout the entire volume, thereby maintaining a constant concentration. This ensures deposition of a uniform fine wetting layer of hydrogen peroxide solution on the target surfaces of the entire enclosure. Unlike the FE method, the surface deposition of droplets does not rely on condensation due by temperature differences. This difference was confirmed by measuring development of the surface film thickness using a laser beam.
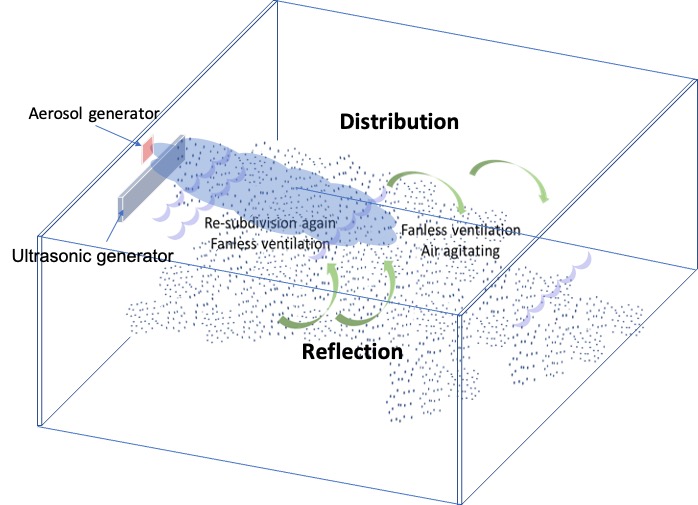
UD method mechanisms
It was also found that the quantity of hydrogen peroxide solution required for effective biodecontamination by the UD method is between 20 and 30% of that required for the FE method. Additionally, it was found that the resulting cycle time, including the aeration phase, could be significantly shortened.
Impact of temperature and relative humidity
A series of experiments were performed inside an isolator to compare the effect of initial temperature and relative humidity on the biodecontamination efficacy for both the FE and UD methods. This experiment was used to define the ‘design space’ that characterises the two methods, and then compare them.
The results are shown in figure 3, and from them the following important findings can be identified:
-
•
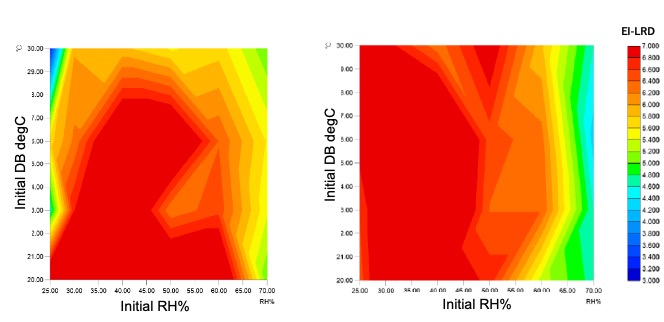
Design Space comparison FE and UD Methods
We can identify the likely reasons for these differences. In case of the FE method, the hydrogen peroxide vapour must reach saturation, enabling micro-condensation to start forming on the target surfaces in the chamber. The inactivation of the EI or BI starts when this wetting layer covers the indicator strip. However, microscopic changes in the extent of this micro-wetting layer, resulting from the complex relationship between temperature, relative humidity, hydrogen peroxide concentration, and airflow limit the efficacy and consistency of the process. Since it is practically impossible to control these intertwined parameters, for example when heat is transferred and the internal air and surface temperature rises, excess hydrogen peroxide is usually added to compensate.
Considering the UD method, the critical and essential difference is that there is no need for airflow or air turbulence inside the chamber to distribute the aerosol. Any unidirectional or non-unidirectional airflow systems are turned off and hence the impact of airflow can be discounted. From these studies it was concluded that with the UD method, repeatable decontamination was possible with a relatively smaller amount of hydrogen peroxide solution, with an initial humidity in the range of 20 to 60% RH and an initial temperature range of 20 to 30°C db.
Airflow velocity impact
It was observed that the higher the air velocity, the greater the biodecontamination effect was for the internal surfaces directly in contact with the outside of isolator chamber; an example being the window area. For the other internal surfaces such as glove surfaces, the EI LRD peaked for air velocities around 0.3 m/s and reduced with increasing air velocity.
This reason for this effect is most likely because condensation of hydrogen peroxide vapour occurs more readily on cooler surfaces in direct contact with the cooler isolator surroundings. This also helps explain why efficacy is frequently found to be poor inside an isolator chamber that uses the classic aseptic processing unidirectional average airflow velocity of 0.45 m/s +/- 20%. The results seen from the cycle development experience also suggest that the optimal unidirectional average airflow velocity for best biodecontamination is typically between 0.25 m/s to 0.35 m/s in the case of the FE method.
On the other hand, when evaluating efficacy for the UD method, the air velocity is zero with the fans stopped and therefore there is no possible influence from internal air flow. This leads to more stable and repeatable biodecontamination being possible without air distribution issues.
Application inside an isolator and irregular surfaces
The biodecontamination intensity distribution for the FE method in an aseptic filling process isolator was investigated. The results showed that the chamber surfaces including the window and the filling machine floor surface achieved stable biodecontamination, but the irregular internal machine surfaces, such as the parts feeder and around the gloves exhibited variations in the biodecontamination potential. Additionally, evidence was obtained from airflow computational fluid dynamics (CFD) to better understand airflow over the irregular surfaces because as identified above, airflow is critical to the performance of the FE method. Figures 4 & 5 clearly show the variable and disturbed airflow over parts of complex process equipment and around isolator gloves.
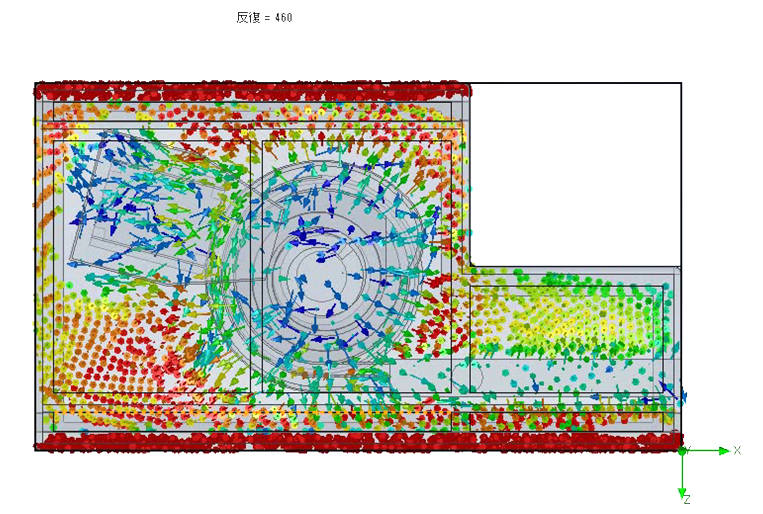
CFD image showing variable and poor airflow around irregular machine surfaces
Biodecontamination of the isolator glove was also investigated (Figure 5). This has an irregular and variable shape and is also difficult to control the internal temperature. The results of these tests were as follows:
1. In case of the FE method, 40 g/min injection of 30% H2O2 solution was required to achieve a robust biodecontamination demonstrated by 6-LRD with EIs around the glove, compared with the UD method, when 12.5 g/min was sufficient.
2. Conversely, when 12.5 g/min injection of H2O2 solution was used with the FE method, a 6-LRD biodecontamination could not be achieved.
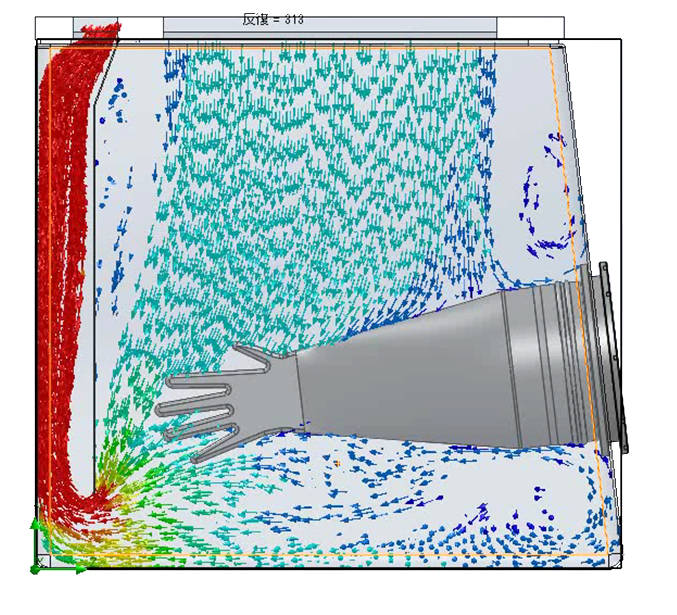
CFD image showing variable and poor airflow over and around isolator manipulation gloves
In other words, the efficacy was improved from 4-LRD to 6-LRD by the ultrasonic wave energy. From these experiments, it was concluded that the UD method can improve biodecontamination efficacy of complex surfaces such as gloves.
Influence of surface materials
It is recognised that for the traditional FE method, the BI D-value (the decimal reduction time) differs greatly depending on the isolator surface materials or BI substrate. Therefore, experiments were carried out to investigate the effect of materials on the efficacy of the UD method. These experiments show the D-value is consistent regardless of the material. This indicates that a stable and repeatable biodecontamination can be achieved. (Figure 6)
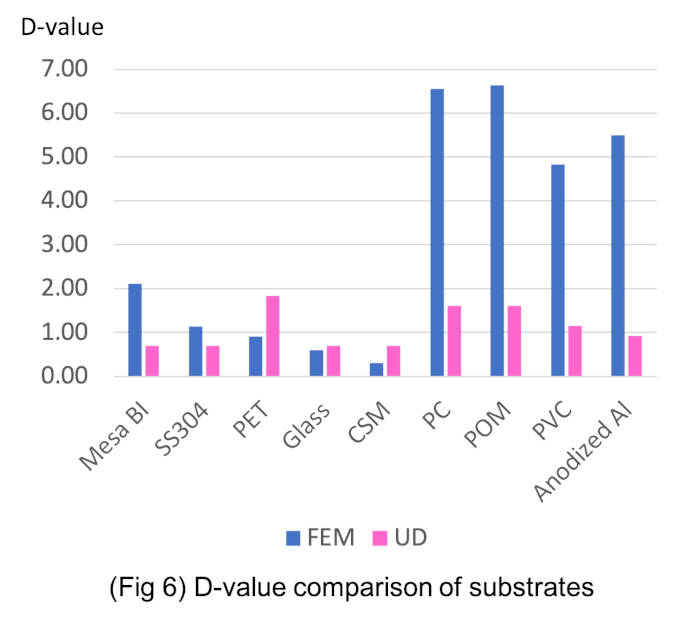
Figure 6: D-value comparison of substrations.
Key to materials substrates:
- Mesa BI: Biological indicator
- SS304: Stainless steel grade 304
- PET: Terephthalate
- Glass: glass vision panel
- CSM: Polyethylene chlorosulfonate (main use glove material)
- PC: Polycarbonate
- POM: Polyoxymethylene
- PVC: Polyvinyl chloride
- Al: Anodized aluminium
Conclusion
This article has presented evidence from experiments and tests demonstrating that the UD method can improve the bio-decontamination robustness and repeatability, shorten cycle time, use a smaller quantity of hydrogen peroxide solution and reduce the impact of environmental variables such as temperature, relative humidity and airflow compared to the FE method. The research and practical testing have also shown the benefit of using EIs, as an adjunct to more traditional BIs. These so-called rapid methods provide the opportunity to obtain more data points more rapidly, enabling more rigorous evaluation of bio-decontamination efficacy.
The fundamental mechanism of the UD method is generation and dispersion of an ultra-fine hydrogen peroxide aerosol to deliver a wetting layer on the target surfaces without requiring internal air movement and avoiding internal temperature rises that can adversely influence condensation of liquid on the target surfaces. Furthermore, the aerosol is maintained sub-divided as a fine droplet aerosol leading to less gravitational settlement, helping to ensure an even spread over inside of the isolator chamber under the influence of the reverberant ultrasonic wave field. Movement of aerosol droplets leads to impingement on the target surfaces of chamber helping ensure that the surface is completely covered by a thin hydrogen peroxide solution layer without requiring condensation by temperature difference. Since the target surfaces are equality covered by the hydrogen peroxide solution layer, robust and reliable biodecontamination can be achieved with a relatively small amount of solution in a short time.
The UD method of bio-decontamination has been used in applications to isolators supplied for the manufacture of ATMPs and traditional aseptically processed medicinal products. It has also been configured and used for room scale bio-decontamination.




