The draft European Good Manufacturing Practice (EU GMP) Annex 1 is the focus of a great deal of discussion. In many ways, this is good as it builds interest, engagement, and inclusion. The downside of this debate, however, is the potential to spread misinformation. It would be prudent for pharmaceutical manufacturers to practice due diligence before changing any procedures.
Let's not forget that the goal of regulatory guidance is to provide standards for basic requirements, not to define procedures. This allows manufacturers to develop and implement procedures that are tailored to their facilities and processes. The procedures must be designed according to each manufacturer’s unique processes, rather than merely adhering to the minimum regulatory standard, as this may or may not be appropriate and can result in misaligned methods. An effective procedure is one that is designed based on a scientific approach and is aligned with the regulatory requirements.
The objective of this article is to clarify when a one or a two-step cleaning and disinfection procedure for cleanrooms and non-contact product surfaces should be implemented. In other words, is a separate and distinct cleaning step always needed before disinfection?
Cleaning and disinfection procedure
“Clean before disinfect” is the golden rule that everyone knows and a legacy from the healthcare and food industry. This principle often relies on bleach, which cannot handle large amounts of soil load and has limited stability. Furthermore, food industry processes can generate high amounts of organic and inorganic residue, which can rapidly deplete products that are not designed to handle such soil. In these cases, the cleaning step aims to remove visible soils from the surfaces, before disinfection, to avoid interference with the disinfectant’s effectiveness. Therefore, a cleaning stage before disinfection is necessary for a manufacturing process that generates a high visible amount of residue.
High levels of residue may be generated in the production of API’s (active pharmaceutical ingredients), tablets or liquid-dose manufacturing cleanrooms where the containment systems may be inadequately designed. In this instance, it is not possible to avoid the accumulation of residues on surfaces.
However, cleanrooms designed with proper containment systems, such as closed manufacturing or hood systems, help to reduce or prevent the build-up of visible residues. Additionally, numerous manufacturing processes generate very low levels of residue. In such cases, is a one-step cleaning and disinfection procedure possible?
The amount of residue generated should not be the only parameter that dictates a cleaning and disinfection programme. The source and nature of the residue should be considered as well. In particular, organic material, for example from a media spill, may present more of a challenge to a formulated disinfectant than light soil from personnel motion (e.g. walking) in highly restricted areas.
It is evident that different parameters must be considered before implementing a cleaning and disinfection procedure. These parameters include:

- Interaction of the residues: If a residue is known to have no adverse interaction with the disinfectant efficacy, then a cleaning step is not necessary. However, if an interaction is known, a separate cleaning step must be performed before disinfecting.
- Amount of the residues: As discussed earlier, a cleaning step is key to removing high amounts of visible material before disinfection. However, in some cases, and depending on the disinfectant composition, a low level of residue may not need to have a separate cleaning step before disinfection (refer to point 3 below).
Note that one can argue how to define a low level (“clean” cleanroom) or high-level (“dirty” cleanroom) amounts of residue. These levels should be decided based on field observations. - Disinfectant composition: The composition of the disinfectant is a key parameter to consider before developing a cleaning and disinfection procedure. Certain disinfectants formulated with surfactants are known to be capable of cleaning and disinfecting in one step. In addition, the mechanical action (e.g. wiping, mopping) using such formulated disinfectants helps to keep surfaces clean. At the same time, the disinfection process is performed by maintaining the wet contact time confirmed during in vitro testing.
Note that the in vitro surface coupon testing separates disinfection from cleaning, focusing on the chemical kill (disinfection) of the product.
According to the US EPA, “an antimicrobial agent identified as a ‘one-step’ cleaner-disinfectant, cleaner-sanitiser, or one intended to be effective in the presence of organic soil, must be tested for efficacy by the appropriate method(s), which have been modified to include a representative organic soil such as 5% blood serum”. However, the EPA also suggested to simulate and confirm the disinfectant’s capability to clean in real conditions where the disinfectant is used. - Disinfectant application technique: Heavily soiled or “dirty” cleanroom surfaces should be cleaned when a disinfectant is applied without mechanical actions such as spraying or vaporising. This technique is applicable even if the disinfectant contains a surfactant.
As discussed earlier, mechanical action using a formulated disinfectant helps to clean while disinfecting. However, a disinfectant without surfactants (e.g. 5% bleach, 3% H2O2) would fall into a grey area, where it may not be as effective for cleaning, even with mechanical action.
Other factors should also be analysed such as the choice of the material of construction of the mop, or wipe that may play a role in removing residue to an acceptable level.
The volume of disinfectant solution being used to mop or wipe may have an impact on disinfectant residue build-up. Therefore, whether the surface is being cleaned and disinfected with a saturated, high-liquid retaining mop/wipe or a flood of disinfectant in an attempt to really wet the area plays a role in residue development. - Environment of use: Disinfectant efficacy studies are typically performed at ambient temperature. However, in cold room processes, the necessary wet-contact times might be as much as 3x higher, depending on temperature and the target microorganisms. In such cases, cleaning before disinfection may help to reduce the bioburden level and may justify shortening the disinfection contact time. In a cold room, the traffic is generally limited and therefore a low frequency of cleaning and disinfection may be implemented, if scientifically justified.
Finally, other factors such as the condition of the area are important to assess, for example, if a floor is damaged and there are areas where product spillage or disinfectant may pool, a higher residue is to be expected.
Scientific justification
The manufacturer should analyse the different parameters examined earlier. The outcome of the evaluation should help to draw a scientific justification for deciding whether a one or two step cleaning and disinfection procedure is required.
The following are two approaches manufacturers may use to determine if a one or two-step cleaning and disinfection process is needed:
- In some cases, for a new facility, or when not enough historical data or observations are available, a simple decision tree may help in determining the cleaning and disinfection procedure (see figure 1).
- When enough historical data or observations are available; a numeric assessment may be possible.
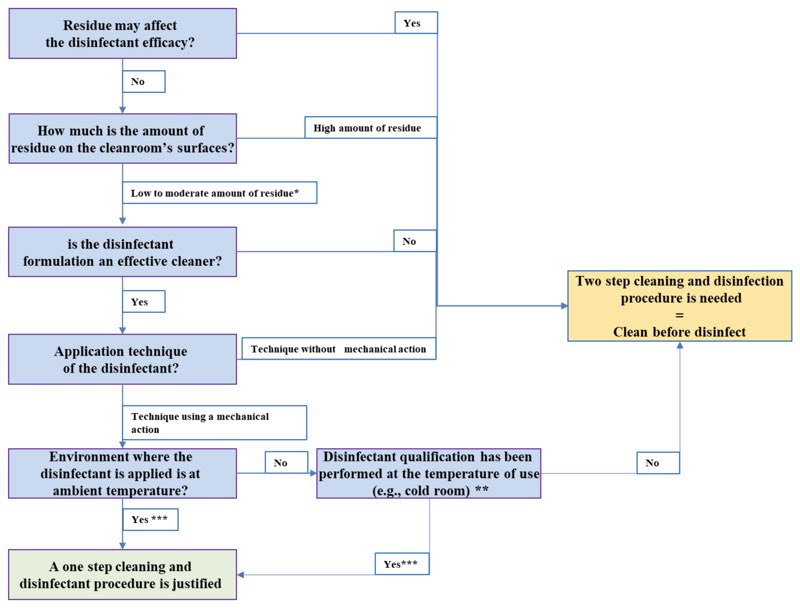
FIGURE 1: DECISION TREE TO JUSTIFY A ONE OR TWO-STEP CLEANING AND DISINFECTION PROCEDURE
* The amount of residue should be assessed based on visual observation of the cleanroom surfaces. Note that external partner with cleanroom expertise may help to define low, moderate or high amount residue.
** If the disinfectant efficacy test on isolate is less than 15 minutes wet contact time, a one-step cleaning and disinfection may be justified. However, if the wet contact time is higher than 15 minutes, it is advised to use a two-step cleaning and disinfection procedure.
*** Assuming the condition of the area is excellent and no damaged of the areas is visible where product spillage, cleaner or disinfectant may pool.
Below is an example of a simple assessment approach, which may help justify a one or two-step cleaning and disinfection procedure: For the sake of simplification, the risk classification used is 1 (low risk), 2 (medium risk) and 3 (high risk) (see table 1).
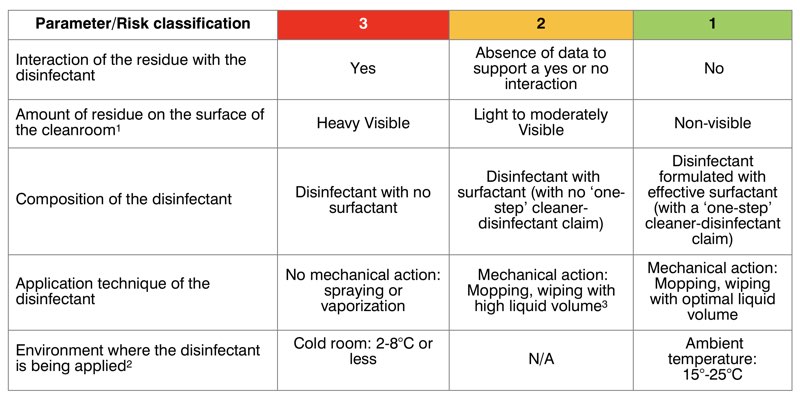
TABLE 1: ASSESSMENT TO JUSTIFY A ONE OR TWO-STEP CLEANING AND DISINFECTION PROCEDURE
* The amount of residue should be assessed based on visual observation of the cleanroom surfaces.
** If the disinfectant efficacy testing has been performed at the expected surface temperature during application, and depending on the wet contact time, the risk number may be reduced or maintained. If the area condition is not optimal, then another factor may be added such as:
Level 3: bad condition of the area (i.e. presence of a crack, colour change), level 2: satisfying condition (minor) and level 1: excellent area condition.
*** The surface being cleaned with a high-liquid volume in an attempt to wet the area would play a role in disinfectant residue emergence.
In this case, the risk priority number (RPN) is equal to the sum of the value of each parameter: lowest RPN equal to 5, a medium RPN equal to 8, and a highest RPN equal to 15. Note that based on historical data and observations, the manufacturer should be able to set up the RPN threshold value. In this example a threshold value is 8:
- RPN higher than 8: a separate cleaning step may be needed before disinfection. This means, even if the disinfectant contains a surfactant in its formulation, a two-step cleaning and disinfection procedure is desirable.
- RPN between 5 and 8, or equal to 8: a separate cleaning step is not required before disinfection. But, if the environment where the disinfectant is applied includes damaged areas where product spillage, cleaner or disinfectant may pool, or includes a cold room —and depending on the microorganism— a cleaning step may be beneficial.
- RPN lower than or equal to 5: a separate cleaning step is not essential before disinfection. A one-step cleaning and disinfection procedure is acceptable.
Effectiveness is paramount
The complete set of factors that affect cleaning and disinfection effectiveness should be identified and considered. An analysis with “in the field” observation is necessary to understand the different factors that impact the effectiveness of the cleaning and disinfection programme.
A thorough analysis of the manufacturer’s surface cleanroom “cleanness”, the technique to apply the disinfectant, the disinfectant composition, and the environment where the disinfectant is used, is crucial to support scientific justification of a one or two-step cleaning and disinfection procedure.
N.B. This article is featured in the March 2019 issue of Cleanroom Technology. The digital edition is available online.

