Designing a vaccine manufacturing facility
Like any other pharmaceutical or biotechnology manufacturing environment, vaccine production requires dedicated cleanroom areas for their vaccine manufacturing facility. As seen in our article designing a CGMP Cleanroom for Cell & Gene Therapy, manufacturing biologic drugs brings challenges to the cleanroom design. Vaccine manufacturing facility design is no exception to this rule.
This article will cover many aspects to help you understand how to design a vaccine manufacturing facility. You will find pieces of information about:
- General cleanroom considerations
- Different types of vaccines
- Vaccine manufacturing facility detailed layout
- Types of equipment needed for vaccine production
- The different steps for manufacturing a viral vector vaccine
- And vaccine-related products manufactured in cleanrooms
All stages of vaccine development and manufacturing need cleanroom environments for delivering the required critical environment. Proper temperature, humidity, airflow, air pressure (positive or negative depending on the BSL level), and other environmental requirements are mandatory to provide a safe vaccine without any risk of contamination.
Here are a few things to take into consideration for a vaccine manufacturing facility or bio-manufacturing facility layout:
- Type of vaccines or components you manufacture
- Biosafety Levels (BSL) depending on the type of vaccines or viral vector used
- Workflow patterns
- ISO classifications / GMP requirements for vaccine cleanroom
- Process stage (R&D, preclinical and clinical stages, large-scale manufacturing)
- Dimensions, equipment inside the room, number of people working inside
- Vaccine production capacity/number of doses expected to output
- Facility expansion
- Standard utilities (plumbing, electricity, process gases, etc.)
You can also have a look at our Ultimate Cleanroom Design Checklist article.
The type of vaccine or components produced inside vaccine manufacturing facilities

We often speak about the vaccine itself and the disease it protects us from. Still, vaccine production requires other components such as proteins, nucleic acid, viral vectors, cells, adjuvants, plasmid DNA, and many more. A lot of these vaccine components need to be manufactured inside cleanrooms. Some vaccine manufacturing facilities will do all the production by themselves or do business with a contract development manufacturing organisation (CDMO) that manufactures specific vaccine components in their cleanrooms.
It’s also important to note that the vaccine manufacturing facility design will change depending on the type of vaccine. A viral vaccine manufacturing facility, for example, will have a different design than a plant-based vaccine or an mRNA vaccine facility because the workflow, equipment, and cleanroom classes will be different.
A cGMP vaccine facility can manufacture:
- Inactivated vaccines
- Live-attenuated vaccines
- Messenger RNA (mRNA) vaccines (Covid-19 vaccines)
- Subunit, recombinant, polysaccharide, virus-like particles (VLP), and conjugate vaccines
- Toxoid Vaccines
- Viral vector vaccines
- DNA vaccines
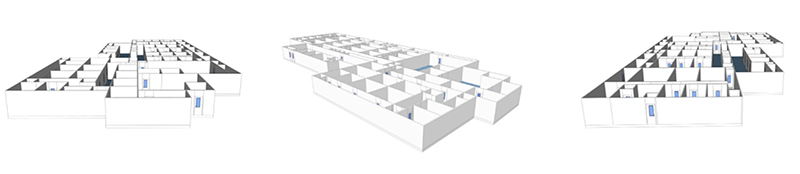
The vaccine manufacturing facility design/layout
Vaccine products range from live organisms (for example, attenuated salmonella typhi) to inert proteins (e.g., virus-like particles used to protect against hepatitis B infection). Such product diversity has a significant impact on facility design, process validation, and control. Here is an example of a viral vaccine manufacturing facility design or layout.
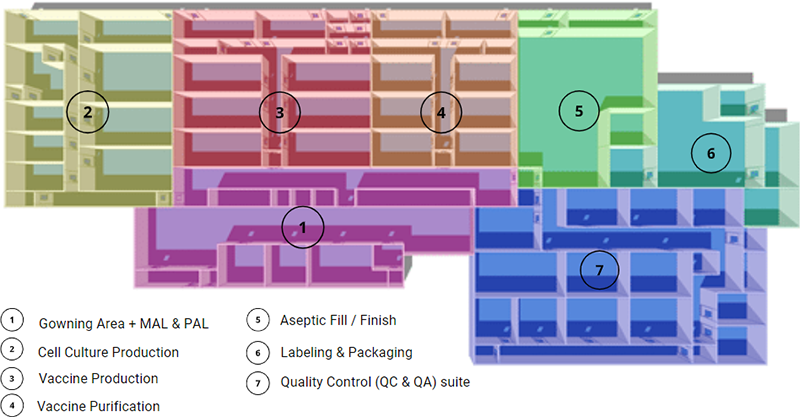
Vaccine manufacturing facility design and layout
- The cleanroom change room / Gowning Area: Like any other cleanroom environment, the vaccine manufacturing facility will need a gowning area to eliminate the risk of contamination. Multiple airlocks will be included in the design, material airlocks (MAL) and personal airlocks (PAL).
- Cell culture production: The removal of cells, tissues, or organs from an animal or plant and places them in an artificial environment to promote growth. The cell culture cleanroom is ideally a segregated area, as you can see in the layout picture above. The cleanroom dimension will be influenced by the quantity, the size of the bioreactors or cell culture fermenters. Their bioprocessing capacity ranges from 50 to 2,000 litres and even more.
- Vaccine production: This is the cleanroom area of the viral vaccine manufacturing facility used to harvest and inactivate the pathogens inside the virus. Suppose you are manufacturing vaccines with viral vectors, depending on the virus strain; you will need to think about the biosafety level (BSL) and current Good Manufacturing Practices (cGMP) requirements in your vaccine plant design and layout. The BSL level will impact the HVAC system and the vaccine facility design, as the cleanroom might need negative pressure for BSL-3 and BSL-4. The cleanroom grade for these vaccine activities is usually a Grade C (ISO7).
- Vaccine purification: Vaccine purification is one of the most challenging tasks of the downstream process as it usually requires multiple purification steps. Technological advances now make it possible to accelerate the purification in the downstream process. The vaccine manufacturing facility will need chromatography equipment for this step. The vaccine formulation, where adjuvants, stabilisers, preservatives are added to the vaccine, will take place in this cleanroom. Environmental monitoring is also something to consider in your design/layout. Thinking through the workflow and manufacturing process is critical when designing your vaccine manufacturing facility.
- Aseptic Fill / Finish: Vaccine manufacturing facility design needs to include a specific area for the aseptic fill/finish for the sterilisation and packaging of the newly produced vaccines. The aseptic fill/finish room is a crucial area of the facility to ensure the vaccine’s safety inside the vials and syringes. In this area, the HVAC will be designed and operated to keep the aseptic core using positive pressure that extends away from the core. Biosafety cabinets or laminar airflow hoods (LAF) will be necessary to obtain an ISO 5 (Grade A) area for critical operations of the filling process, with an ISO 8 (Grade D) cleanroom background.
- Labelling and packaging: Even though this area doesn’t need any particular cleanroom grade or ISO class, it is a substantial part of the vaccine manufacturing facility design. It is crucial to ensure that the suitable vaccine will go in the correct vial with the right label. A Clean Non-Classified (CNC) or Unclassified (UNC) cleanroom grade is respecting the World Health Organization guidelines, depending on if the vaccine capping is done or not. Usually, the capping would be done in the previous phase of the vaccine production. The vaccine production plant will need packaging, vial labelling machines, and cold storage equipment such as freezers (the newer vaccines need to be kept at -70°C/- 94°F).
- Quality Control (QC & QA) suite: The quality control suite is essential for testing the potency of the vaccines being produced. Living organisms and biological drugs are way more complex than chemical drugs. The reaction of the living organisms inside the vials may differ in every batch of the vaccine production, and it’s why they need to be tested carefully. In an open area, the quality control lab needs a cleanroom grade A in a Grade B background. In a segregated and dedicated QC lab, an isolator with a grade D background will work.
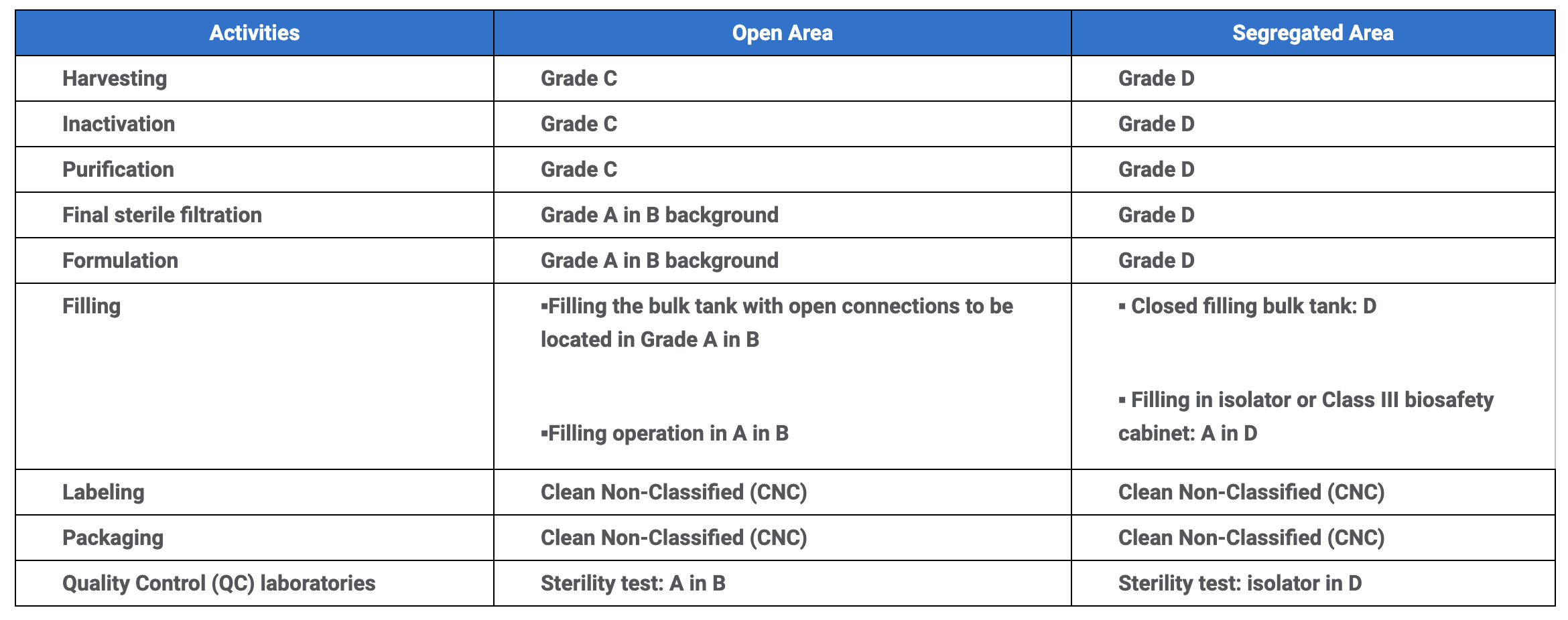
GMP cleanroom grades for vaccine production activities
*Source: https://www.who.int/immunization_standards/vaccine_quality/env_monitoring_cleanrooms_final.pdf
**This table is for information purposes only. Regulations may change depending on the application.
Vaccine stakeholders: from cleanroom environments to large-scale vaccine manufacturing
We often think of vaccines as the end product of a pharmaceutical company, but the truth is that vaccines are the result of mankind’s knowledge and collaboration. Developing vaccine candidates as fast as we did for the COVID-19 pandemic requires various expertise and specific skills. This was all possible due to the involvement of various specialised vaccine-related companies and their products. There are multiple stakeholders involved from the development of a vaccine to its commercialisation. The stakeholders range from medical device companies to biotechcompanies to the pharma industry.
The life sciences field is evolving quite rapidly thanks to new technologies and further understanding of the human body. Vaccines are usually known for boosting immunity against infectious diseases. Still, today they are also used in immunotherapy with CAR-T cell vaccines, and many vaccine technologies are used in delivering monoclonal antibody injections for autoimmune diseases. Many vaccine components are also used in other types of drugs too. In other words, vaccine manufacturing facilities and vaccine-related cleanrooms are used to manufacture diverse biological drugs.As there are so many different vaccine-related products, the manufacturing process can vary for many reasons, which means that the cleanroom layout becomes a unique design.
Here are a few examples of the different stakeholders’ products that are involved in vaccine production:

Vaccine related products
Equipment list for vaccine manufacturing facility
Vaccine equipment varies depending on the type of vaccines produced and the size of your operations, whether you are in the development phase, preclinical stage, or large-scale manufacturing. Here is a summary list of equipment and cleanroom design considerations for your vaccine process and layout.

