What can drug manufacturing companies do to become more sustainable and how can technology providers help them to achieve their ambitious goals? A very important lever lies in calculating the carbon emissions of machines, as current analyses show.
The pharmaceutical industry is a sector of extremes. New drugs are dramatically increasing patients’ life expectancy, prices for ground-breaking therapies are exploding – and, unfortunately, resource consumption is still considerably exceeding that of other industries.
Technology providers not only need to fulfil their own emission targets; they are also indirectly responsible for those of their customers
Way before the current rise in energy prices, the sustainable use of energy and resources became a major challenge for pharmaceutical manufacturers. The industry counts around 20,000 companies worldwide, and their impact in drug sales and carbon emissions is considerable. According to a study by McMaster University, the pharmaceutical industry produced around 50 metric tonnes of carbon dioxide equivalent (CO2e) per million US dollar sales in 2015 while total sales amount to $962 billion globally – even more than the worldwide automotive industry.
High cost and sustainability pressure
Despite all criticism, the pharmaceutical industry is also under great pressure: life-saving medicines must reach the market in high quality and within specific timeframes. At the same time, drug manufacturers are supposed to design processes that generate significantly fewer emissions.
This balancing act calls for lower-emission technologies, which have set off a race for innovative strategies in the industry. Eighty per cent of the largest companies in the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) want to reduce their emissions as far as possible or even achieve CO2 neutrality, while some key market players are aiming even higher.
This also impacts the players in the second or third line: contract manufacturers and technology providers. Scope 1 emissions of pharmaceutical manufacturing companies include so-called “direct” emissions, i.e. those caused by operating things they own or control. According to the World Economic Forum, direct emissions “can be a result of operating machinery to make products, drive vehicles, or just heat buildings and powering computers.” Scope 2 deals with energy purchased by the company itself, while Scope 3 includes other indirect emissions related to a company's value chain, and which can be caused by business travels, commuting, waste disposal, or the use of sold products.
Hence, technology providers not only need to fulfil their own emission targets; they are also indirectly responsible for those of their customers.
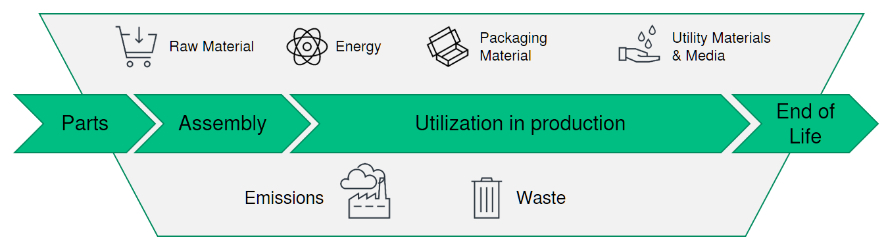
The role of technology providers
While drug manufacturing companies are pursuing different strategies and have set themselves different goals with varying timelines, they do share a common vision – and need their partners to support them in their sustainability endeavours. Calculations of carbon footprints, science-based targets, or sustainability reporting are on the agendas of pharmaceutical producers, contract manufacturers and technology providers alike. The latter play a key role in the pharmaceutical industry’s sustainability strategy since equipment emissions are a major part of the carbon footprints of both CDMOs and drug manufacturers.

Due to its typically long lifespan, equipment usually contributes to overall emissions over several decades. One of the biggest levers for CO2 reduction therefore lies with the technology providers themselves: by continuously optimising their machines and developing new, more energy-efficient solutions, they can contribute significantly to lower emissions and more sustainability in the entire industry.
Downstream processes also offer savings potential, especially through innovative, alternative packaging materials. Liquid pharmaceuticals require glass and single-use plastic in primary packaging, leaving hardly any room for change. However, recyclable and/or biodegradable solutions are playing an increasingly important role in the primary and secondary packaging of solid dosage forms. Blister packs made of paper or recyclable polypropylene (PP), which aim at substituting standard thermoforming films made of polyvinyl chloride (PVC), can be used for the primary packaging of tablets and capsules. Secondary packaging in turn largely depends on cartons.
Disclosing the carbon footprint
Along with the technology, its market price and total cost of ownership (TCO), the equipment’s emissions have become crucial for purchasing decisions. By disclosing their own emissions and carbon footprint on platforms such as CDP or the Science Based Targets Initiative (SBTi), equipment manufacturers increase transparency and set clear, publicly available targets for improvement.
Assessments by agencies such as EcoVadis, which rate companies on various sustainability criteria, confirm that they act sustainably and in accordance with international standards.
A further field of action for technology suppliers includes software-based CO2 analyses. New methods are available to analyse the carbon emissions along the entire life cycle of the equipment with so-called Life Cycle Assessments (LCA). Such evaluations serve a double purpose: machine manufacturers themselves can use the data to identify potential for future technological optimisations at an early stage.
The equipment’s emissions have become crucial for purchasing decisions
Pharmaceutical manufacturers, in turn, receive the data they need to make targeted investment decisions for choosing process alternatives or equipment that helps achieve their own sustainability goals.
Knowing the carbon footprint of a machine can provide orientation and support decision-making, e.g. for alternative production processes. For example, pharmaceutical companies can use both a membrane-based and a distillation process to produce WFI (water for injection) for parenteral applications.
Analyses show that “cold” WFI units produce up to 90% fewer CO2 emissions than the established “hot” processes. Even WFI from cold processes that use hot storage reduces emissions by more than 40 per cent compared to hot WFI.
Sterilisation tunnels as an example
The most important parameters of the footprint calculation include released thermal energy, electrical power, compressed air, and other media. A profound analysis, however, needs to take many more parameters into account. They include the average service life of the equipment, but also the times for commissioning, heating up tools, and performing maintenance activities, as well as downtimes.
Typically more than 90% of all carbon emissions of the sterilisation tunnel occur during usage
Moreover, the impact of packaging materials on emissions can also be calculated.
One of the most energy-intensive processes in the pharmaceutical industry is use of sterilisation tunnels. They are crucial for maintaining the sterility of containers and significantly contribute to product safety. According to LCA analyses, typically more than 90% of all carbon emissions of the sterilisation tunnel occur during usage. Materials rank second highest, while the equipment’s end-of-life amounts to less than one per cent of the emissions during the machine’s entire life cycle. This data not only helps pharmaceutical manufacturers to see where they can save energy. It also provides the basis for concrete recommendations from technology providers, as the following example illustrates.
Up to 10% electricity savings
Depending on its operating state, the sterilisation tunnel uses different amounts of energy. This provides several levers for more ecological operations without compromising quality or validation. With a length of several meters, the tunnel consumes up to 15 per cent more energy during heating than during operation. Changing the operating state offers potential for savings: many drug manufacturing companies keep their tunnels ready for operation, i. e. in high-level standby mode, for very long periods of time to reduce heating energy – even if they only use them for a few hours per day. They only switch to low-level standby with less power consumption on weekends.
This operating mode ensures sterile conditions at a temperature of 130 degrees Celsius. However, the tunnel must be heated up to 350 degrees Celsius again for regular production. LCA analyses show that low-level standby is also possible during weekday downtimes without jeopardising sterilisation quality. Compared to high-level standby, manufacturing companies can save up to ten per cent of electricity – without having to requalify the system. This specific example helps pharmaceutical manufacturers to rethink their operating strategy in view of more sustainable processes. The entire LCA analysis also provides important data for the equipment manufacturer, which can be used to optimise its own technology in the long term.
The foundations for sustainability in pharma
Considering the current developments and endeavours, the pharmaceutical industry might achieve climate neutrality within the next two decades – at least those companies that are already pursuing a clear sustainability strategy. In the long term, this trend will include other players besides “big pharma”, such as contract manufacturers and smaller manufacturing organisations. Media and energy consumption as well as avoiding waste in production, transport, and packaging will be among the main levers.
Moreover, software updates for existing machines and CO2 analyses can form the basis for more energy-efficient operations.
Technology providers who have already invested in their own sustainability strategy at an early stage can now position themselves as valuable partners with additional energy-saving options and attractive new technological solutions.
