By adjusting cleanroom airflow based on environmental factors, the room solely receives the volume of air required to maintain critical parameters within compliant levels – namely particle concentration, microbial contamination, temperature, humidity and differential pressure. In case of a contamination event, the system is able to rapidly increase airflow to offset this rise in contamination and continuously guarantee a compliant environment for operation.
This approach ensures a compliant cleanroom environment at all times, which is especially valuable during high-emission processes such as room cleaning or the operation of equipment.
Interestingly, when compared to a static cleanroom, a dynamic space is able to deal with contamination in a more effective manner, seeing a significantly shorter recovery time than that of a cleanroom utilising a fixed air change rate.
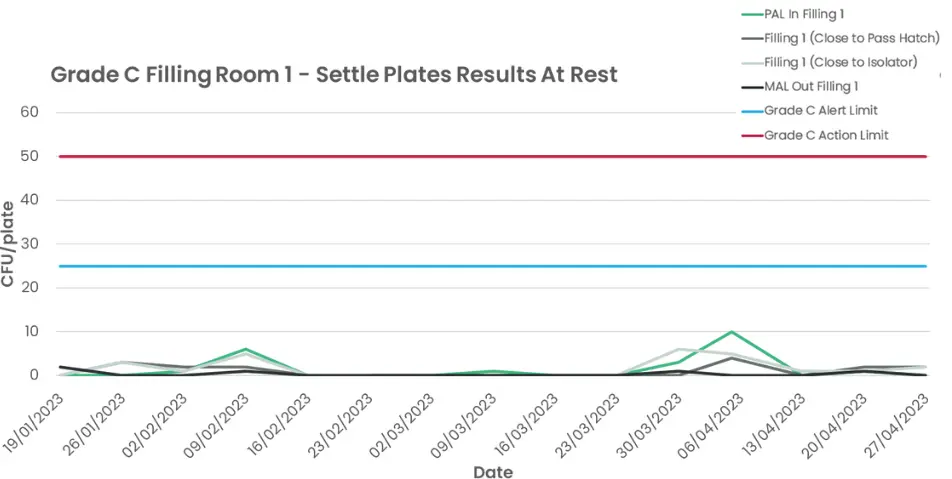
Viable contamination counts during dynamic airflow over a period of four months.
As can be seen from the graph above (Figure 2), the cleanroom space experienced only small levels of viable contamination during a four-month period in which demand-based airflow was constantly active in the facility. The data within Figure 1 is also encouraging, as it shows that in spite of a contamination challenge being introduced by the act of seven operatives cleaning the isolator, the system was still able to control the particle concentration to remain below 10% of the compliance limit for the classification.
How do you qualify a cleanroom with dynamic air change rates?
The approach to qualification follows a similar method to that of a typical cleanroom using a fixed air change rate. This involves qualifying the space at a selected low supply air rate, which for Cambridge Pharma’s sterile fill finish (EU GMP Grade C) cleanroom is 5 air changes per hour in the “at rest” condition. During performance qualification (PQ) the cleanroom is closely monitored under various ‘worst-case’ scenarios and other potential operating conditions to assure the quality of the environment.
How is room differential pressure affected by dynamic airflow in a cleanroom?
Controlling the supply and return or extract air flow rate of a cleanroom space is essential to maintaining room differential pressure. When air is introduced at an increased rate (as a response to increased particle concentration), the variable air volume (VAV) system will adapt to remove a similar quantity of air from the space, thus preserving the existing differential pressure regime. The use of VAVs allows for highly precise attenuation of airflow and thus is a key enabling technology within a dynamic cleanroom control system such as ICCS®. Other technologies, such as interlocking systems that only allow for the opening of a single door at a time, are also crucial to ensuring correct room pressurisation.
Where does dynamic cleanroom control sit within a contamination control strategy as outlined in Annex 1?
Annex 1 develops the concept of a Contamination Control Strategy (CCS) further than any prior GMP documentation. In this regard, Annex 1 specifically references the use of monitoring systems within cleanrooms to “optimise the detection of environmental contamination”, sections 2.3 & 2.5. As the ICCS® continuously monitors critical parameters (such as contamination) to understand the amount of airflow required to maintain a quality environment, it can be said that this technology is closely aligned with the concept of a Contamination Control Strategy as established in Annex 1.
Current GMP regulation such as Annex 1 further supports the deployment of dynamic cleanroom control technology. As stated in section 2.1 “The use of appropriate technology should be considered to increase the protection of the product… and assist in the rapid detection of potential contaminants”. By employing ICCS® in a cleanroom facility, it is possible to quickly manage and abate contamination levels thanks to the use of real-time data informing the system.
How much energy can a dynamic cleanroom control system save?
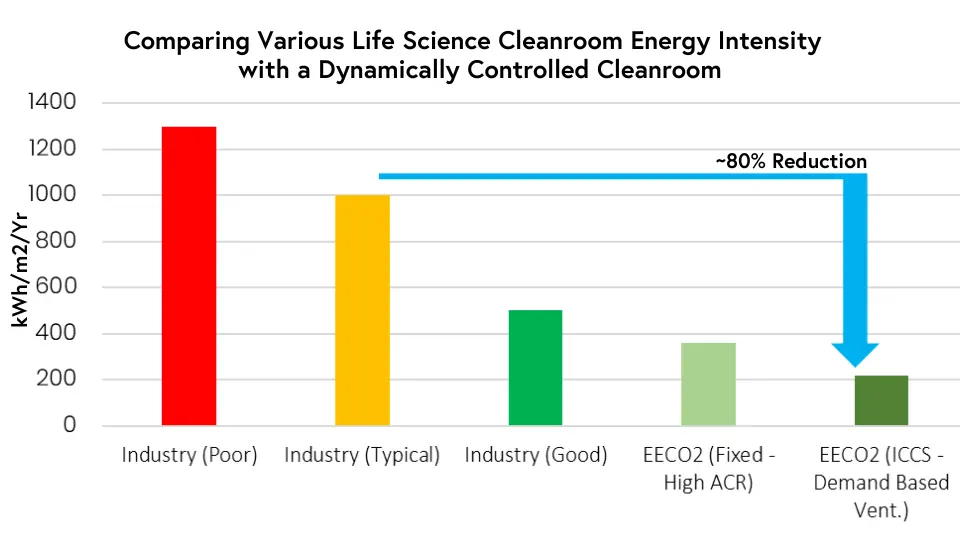
In our experience at over 300 life science sites around the globe, the Cambridge Pharma facility represents one of the most energy-efficient cleanroom spaces in the world. Consuming just over 200kWh per metre squared a year, the new facility has benefited from a combination of energy-efficient design alongside dynamic demand-based airflow provided by ICCS®. Ultimately, this has resulted in an 80% reduction in energy consumption when compared with a typical life science cleanroom space.
Want to learn more about dynamic cleanroom control? Take a look at our exploratory article with Cleanroom Technology HERE.




