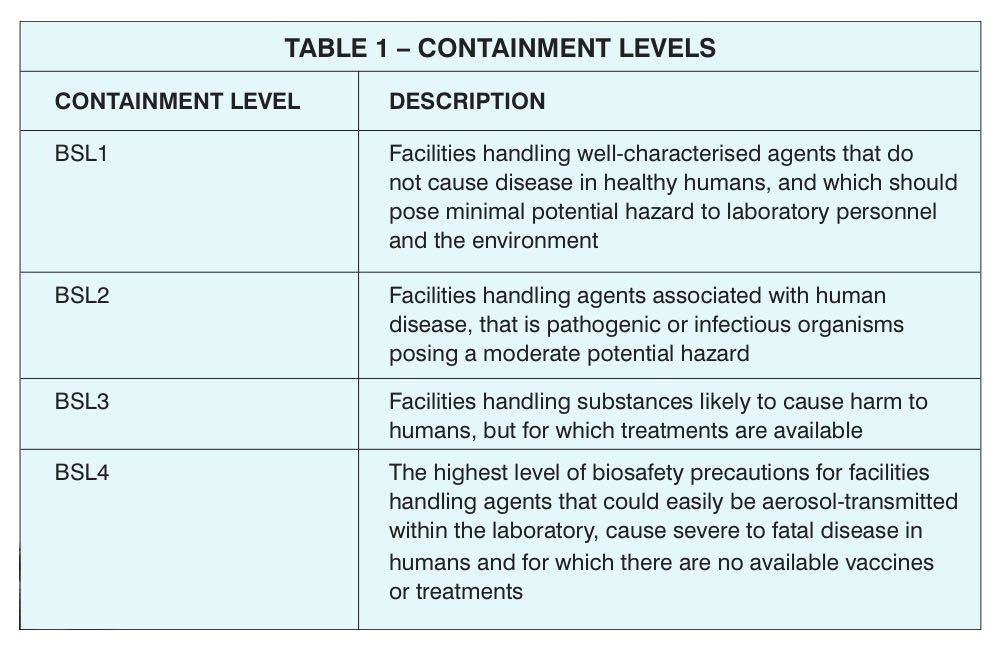
Microbiological containment laboratories across Europe (government, healthcare or pharmaceutical production facilities) must meet the requirements of the Biological Agents Directive, and in the UK this means complying with the Control of Substances Hazardous to Health (COSHH) Regulations. For those unfamiliar with containment levels, these are summarised in Table 1.
Whatever containment level is in force, there is always a potential risk for release of active agents into the environment. Since microorganisms can easily find their way into waste liquids, decontamination of effluent prior to discharge is crucial.
Making your effluent safe
Effluents can include sanitary waste from toilets, water from sinks and washing facilities, and any unsterile exhaust water from sterilisers and process equipment within the containment area. They can contain significant levels of pathogens, and effluent decontamination systems (EDS) are designed to treat them to ensure that they are rendered safe.
Legislation or codes of practice may demand an EDS, or it may be required as the result of an internal risk assessment. However, many ethical organisations install one simply to demonstrate to their neighbours that their operations are safe, and to promote community support.
Pathogens can be killed by a variety of chemical disinfectants such as chlorine dioxide, sodium hypochlorite, hydrogen peroxide and a range of organic chemicals, many of which are highly toxic. These chemicals can present a hazard to operators and, should they be released into the environment, to wildlife and the general public.
For companies to be certain that effluent is safe, it is necessary to ensure there is excess disinfectant remaining in the wastewater after the requisite contact time, which is dependent on the chemical being used. But the excess disinfectant then has to be neutralised prior to discharge of the wastewater to prevent possible environmental impact.
This process means careful chemical monitoring—not always straightforward—and control. Chemical disinfectants are effective against bacteria only if they can reach the cell wall. Solids in the effluent can shield bacteria from the disinfectant chemical, so this also renders it ineffective.
The best alternative to chemical disinfection is heat sterilisation. Heat has long been used for microbiological control in the food, pharmaceutical and healthcare industries.
Pasteurisation at 80°C kills most common bacteria while sterilisation at 121°C or 132°C destroys all forms of life, including viruses, bacteria, fungi and spore forms. Because all the effluent is heated uniformly—including any solids content—there is no escape for the pathogenic organisms.
How it works
EDS systems are usually batch processed, using one or more jacketed pressure vessels or “kill tanks” heated by high-pressure steam. Where possible EDS systems are located in a basement so that the kill tanks can be gravity fed.
Where this is not possible, pumped systems—where the incoming effluent is fed to a holding or storage tank and pumped to the kill tanks—can be specially designed to suit any specific site conditions.
Liquid waste is fed into the kill tank until the level reaches a pre-set point, which starts the heating/sterilisation cycle, and in a multi-tank system, the incoming flow is diverted to another tank until it too is at the correct level to begin the process.
For continuous high-flow rates in the range of 1000 L/h, it may be that a batch decontamination system is impractical or uneconomic. In these cases, a continuous flow EDS system may be the answer
The temperature of the kill tank contents is raised to 121°C or 135°C, depending on circumstances, and maintained for a preset time to effect sterilisation. The sterilised effluent is then cooled to the allowable discharge temperature (normally 60°C) before the contents of the tank are discharged under gravity or pumped away.
For systems where the effluent flow is unpredictable, a holding tank may be required. For continuous high-flow rates in the range of 1000 L/h, it may be that a batch decontamination system is impractical or uneconomic. In these cases, a continuous flow EDS system may be the answer: these systems don’t have kill tanks as such. Liquid waste is collected in a buffer tank, to allow for variations in the incoming flow rate, and then pumped at a constant flow through a series of heat exchangers.
The first pre-heats the effluent using the hot treated effluent and recovers up to 80% of the heat energy.
The second heat exchanger raises the temperature to 140°C, normally using pressurised steam, with a residence time of one minute to provide sterilisation.
Finally, a third heat exchanger cools the effluent leaving the pre-heater to discharge temperature using either air or cooling water. However, if solids are present in the liquid, they may foul the heat exchangers and block the relatively small bore pipework in continuous systems, so batch processing is more suitable for effluents that contain solids.
A reliable tool
Temperature is a simple and reliable online measurement that can be readily calibrated against a standard thermometer and, of course, heat decontamination is completely chemical-free. This method makes control and validation much easier than with chemical systems. Given the high cost of many chemical disinfectants, heat sterilisation is also more cost-effective.
While the temperature in an EDS system is constantly monitored throughout the process, validation protocols require proof that the required level of decontamination is achieved. This is usually carried out by injecting a known solution of B Geostearothermophilus spores at the start of the process and then taking samples from the discharge for laboratory testing. This will give the confidence that the process is effective.

The best EDS solution is a bespoke system
EDS units are critical to the safe operation of a contained facility, so it is imperative that they are constructed to the highest standards of quality and reliability. Heat sterilisation is straightforward enough, but every facility is different in such aspects as layout and space, utility availability, and effluent volume. The pattern of production is also unique in terms of flow rate, frequency and duration.
For these reasons, most EDS systems are bespoke to suit the application, and the number and size of the vessels and tanks optimised.
The engineering is deceptively simple and systems should be designed, installed and regularly serviced by experienced engineers. They should be familiar not only with typical electrical and drainage connections but also have experience of installation connections coming from a high containment facility and expertise in project management.
N.B. This article is featured in the August 2019 issue of Cleanroom Technology. The latest digital edition is available online.
